OUR RESEARCH
viral Host ranges and antibody immunity
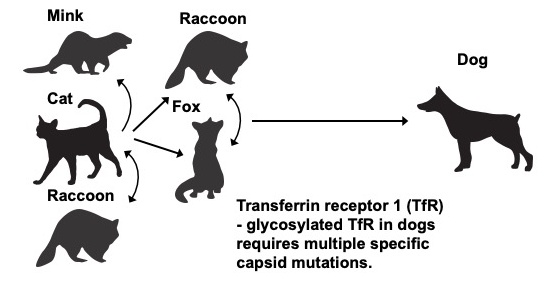
We are been working on understanding the controls of viral host ranges by studying three systems where viruses jumped from one host to another to cause epidemics of disease. Canine parvovirus (CPV) transferred from a cat or closely related host. The H3N8 canine influenza virus transferred from horses, while the H3N2 canine influenza virus transferred from an avian host. The studies include a variety of analyses of the viruses, their interactions with their hosts and environments, as well as their natural histories, epidemiology, and the ecology of their original and new hosts.
Virus Evolution
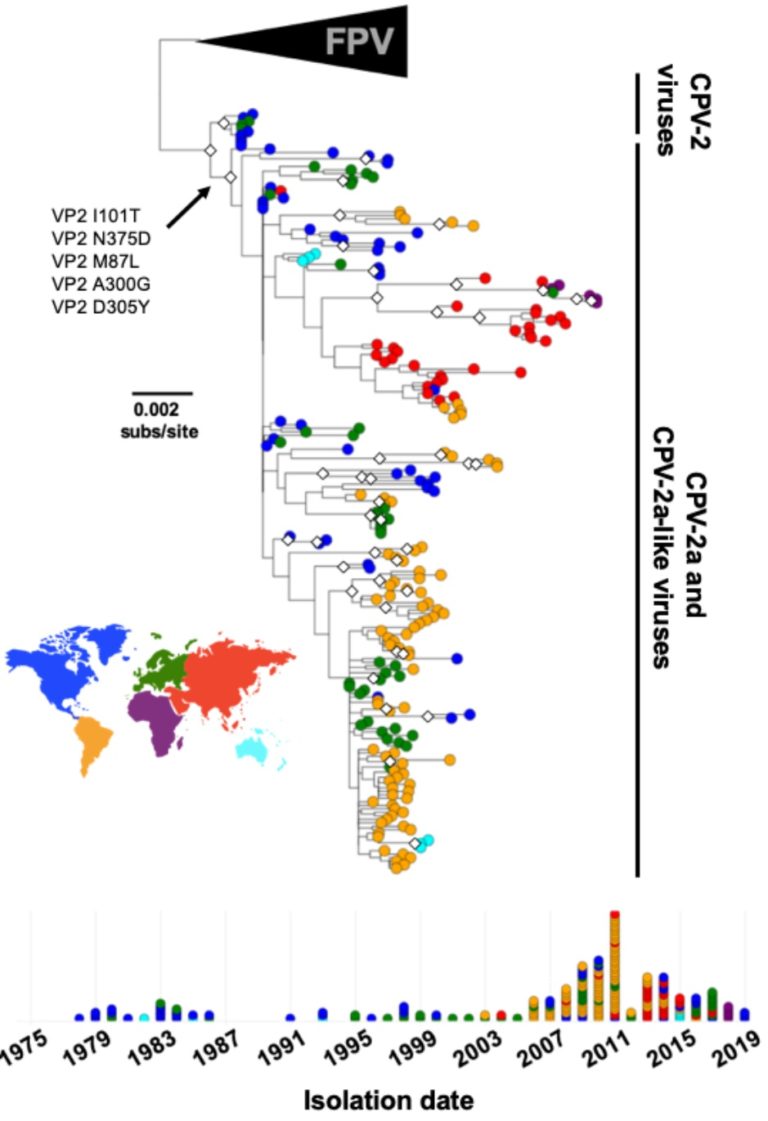
Our studies in the area of virus evolution are focusing on the evolution of CPV, and the two canine influenza viruses – both the H3N2 and H3N8 variants. We are examining the full genomes and deep sequences of the viruses since the earliest points in the viral spread in their new hosts. We are also examining the comparative evolution of the viruses in their original hosts – cats and other animals for CPV, and horses for the H3N8 canine influenza viruses.
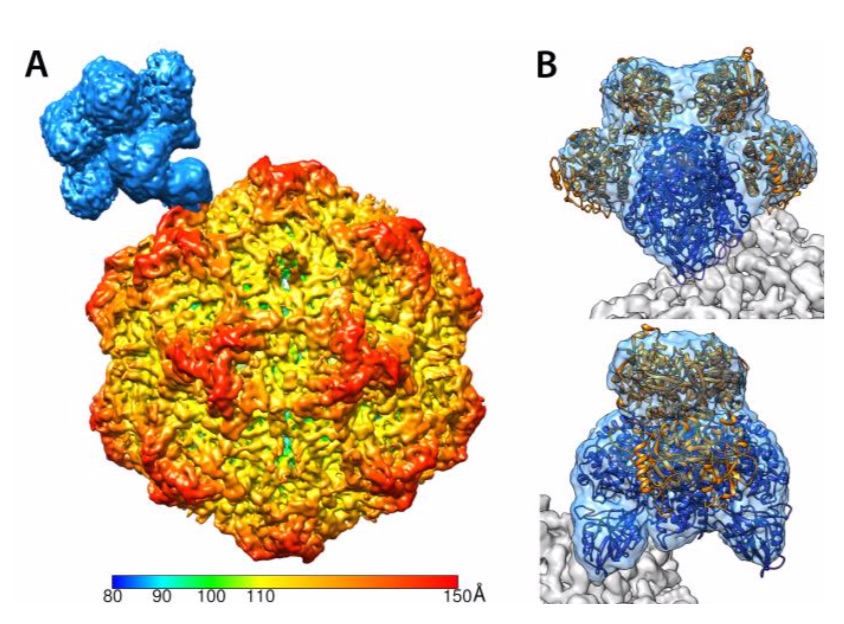
Structures - of virus, receptors and antibodies
We are examining the structures of the viruses and of host components that they interact with – the transferrin receptor type-1 (TfR), antibodies, and sialic acids. This work started with the X-ray crystal structures of canine parvovirus (CPV) and feline panleukopenia virus (FPV), and has more recently included cryoEM analysis of the TfR in complex with the CPV capsid, and antibody:capsid complexes.
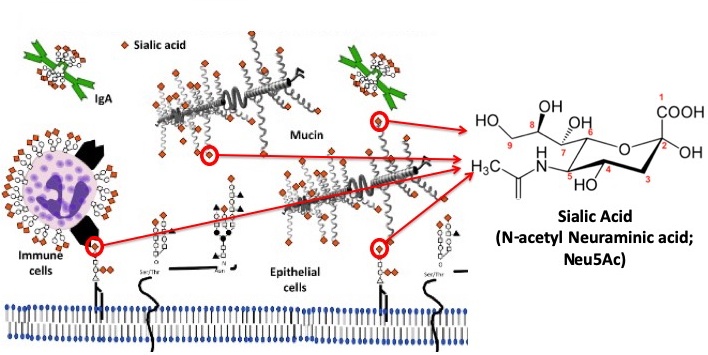
Sialic acids and mucus
Sialic acids are the receptors for many viruses, and are also components of the mucus that covers all mucosal surfaces, in saliva, and on glycans (glycoproteins and glycolipids) of cells and tissues. We are examining the sialic acid forms present in the tissues of different animals, and their effects on binding and entry of viruses, bacteria and parasites, and effects on enzyme activities (e.g. neuraminidases).
OUR COLLABORATORS
We have many collaborators, including Susan Hafenstein’s lab at Penn State University, Ajit Varki’s lab at UC San Diego, Kirk Bergstrom’s lab at UBC Okanagan, Rachel Willand-Charnley’s lab at South Dakota State.
FUNDING AND SUPPORT
The research in the laboratory has primarily been supported by the National Institutes of Allergy and Infectious Diseases, of the NIH. 